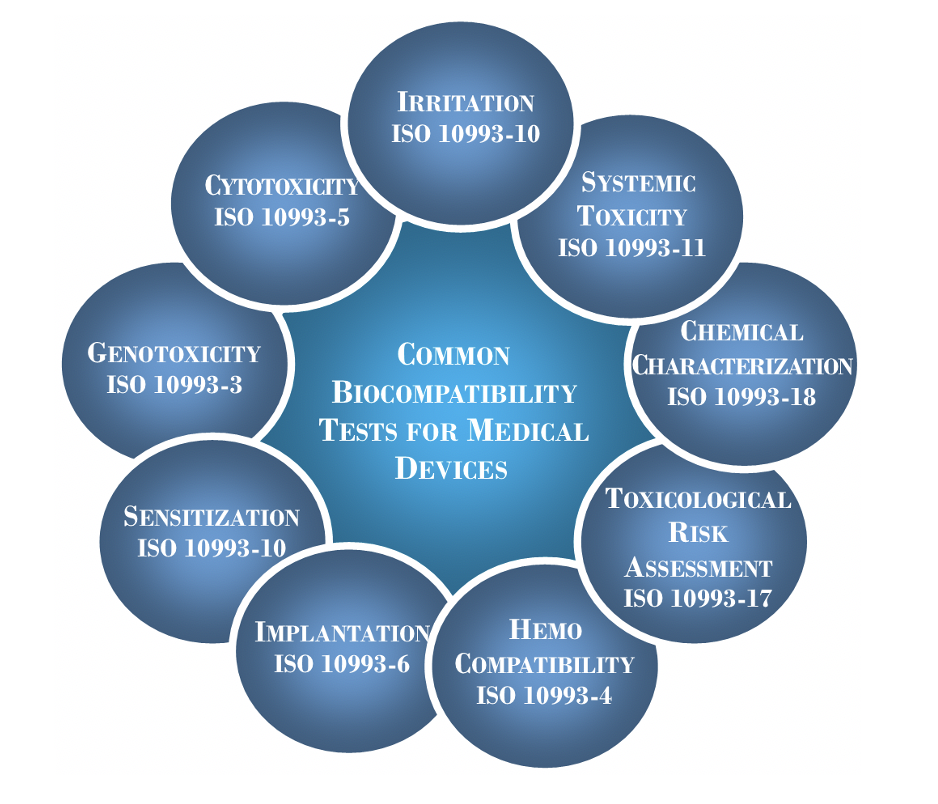
ISO 10993-1:2018 is the overarching biocompatibility guidance document, which emphasizes medical device patient contact duration and contact type (i.e. tissue contacting, blood contacting, externally communicating, etc.) to determine biological risk.
Our testing program uses this standard to guide which biological tests are necessary for your device dependent upon the device contact and duration of use. These may include a number of different biological endpoints.
Utilizing the concepts and principles outlined in ISO 10993-1:2018, a comprehensive and custom biocompatibility test plan is created dependent upon your unique medical device classification.
Once all biological endpoint testing has been conducted, a comprehensive biological risk assessment is created that references all ISO 10993 testing, along with ISO 18562 (if applicable) in order to support 510(k) submissions, notified body submissions, post-market product changes, and/or new product development.
ISO 10993-17:2002, ISO/TS 21726:2019
The toxicological risk assessment is a critical component of a comprehensive biological risk assessment. This analysis utilizes concepts to include health-based safety thresholds, tolerable intakes, tolerable exposures, and threshold of toxicological concern (TTC) in order to derive a risk estimation and hazard characterization of either leachates, extractables, or volatile emissions from a medical device.
All toxicological risk assessments are customized to the type of device, patient use case, and patient age/weight in order to give an accurate, yet conservative estimate of toxicological risk that may prompt further biological evaluation of the medical device, or be used in conjunction with other biological endpoints for a comprehensive biological risk assessment.
ISO 10993-18:2020
Key inputs into the Toxicological Risk Assessment of a medical device include Chemical Characterization after Extractables and Leachables Testing. The incorporation of additives to suit device applications often includes plasticizers, stabilizers, colorants and processing aids, all of which can impact patient safety. Extractables and Leachables testing enables the determination and quantification of chemical compounds that may leach and /or extract out of a medical device during use. This in turn can inform the overall biological risk assessment of the device and its impact on patient safety, including if additional biological testing is warranted.
PSN has developed an advanced process for Extractables and Leachables Testing, which is offered under our ISO/IEC 17025:2017 accreditation scope. Our scientists and SMEs actively monitor FDA feedback through published literature to incorporate a revised approach.
PSN’s toxicology team works in harmony with the analytical team to deliver the final report with a toxicological risk assessment